 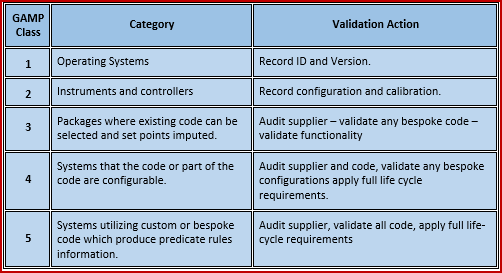
By playing an active and coaching role, we provide validated systems that satisfy every aspect of compliance. In short, we offer GAMP 5® consultants with the right experience and skills for short and long-term projects – internal or external. If you are not sure if your system needs to be validated, we advise you on the best approach and working method. We are able to guide your company during validation processes, or completely carry out your project from A to Z, including planning, testing and writing documents like user requirements. QbD offers consultants for various kinds of assignments. GAMP 5 should be implemented for the automated systems in pharmaceutical manufacturing and quality control to produce the high quality. Together, we tackle possible gaps to make your quality system more robust and audit-proof. The new GAMP-5 guidelines were released February at the ISPE Manufacturing Excellence Conference in Tampa, Florida. We work in the most efficient way possible and we help you focus on product quality and patient safety while reducing validation costs. 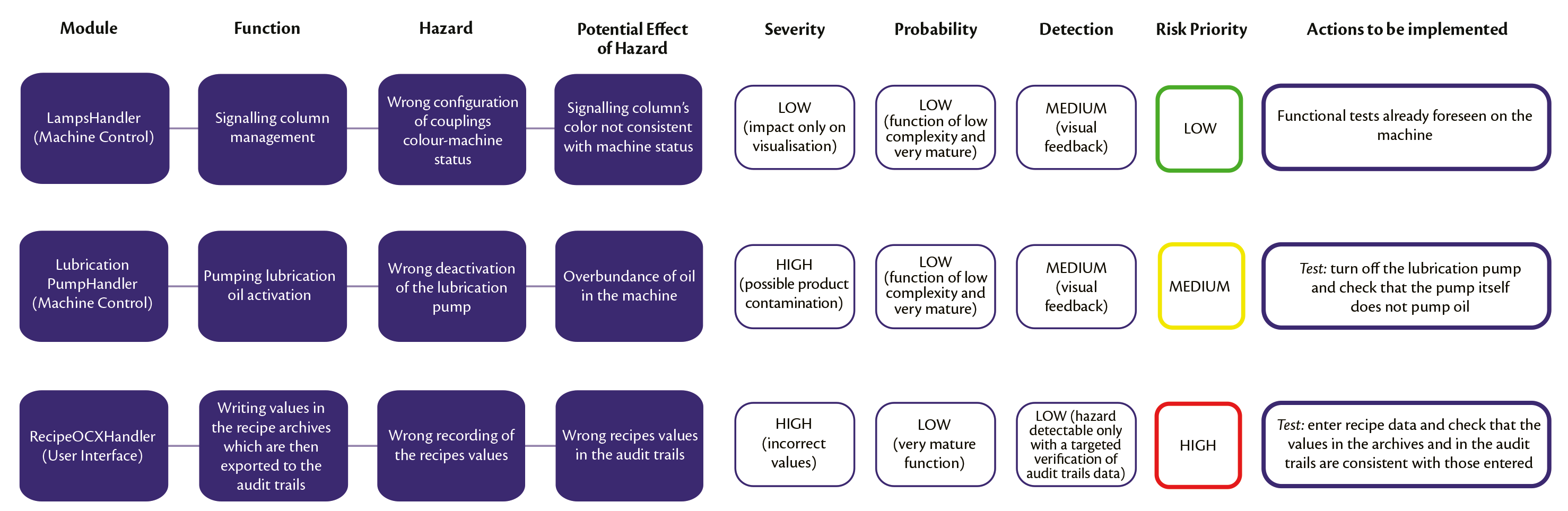
Our GAMP 5® experienced consultants are up to date on the latest insights (including Data Integrity, outsourced systems, artificial intelligence and machine learning) and make use of a pragmatic, risk-based approach – based on their expertise and industry best practices. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
